Energy Harvesting Versus Batteries for Multidecade IoT Sensor Operation
Contributed By DigiKey's North American Editors
2019-12-03
The availability of ultra-low-power ICs has led to a proliferation of sensor-based circuits and systems which require only microamps or milliamps for operation. These products are used for environmental monitoring (safety, temperature, earthquakes), asset tracking, energy/power metering, and medical monitors, as a few examples.
Many of these units need to function for ten, twenty, or more years, yet it is often difficult or impossible to access them for battery replacement. Changing batteries is also often cost prohibitive.
As a result, designers must give due consideration and decide among options, or a combination of options, that include:
- Energy harvesting
- A rechargeable (secondary) battery with energy harvesting
- Ultra-long-life non-rechargeable (primary) battery
- A supercapacitor with ultra-long-life primary battery
This article will look at the various options available to a designer and their trade-offs. In the process, it will introduce related solutions and their use from Tadiran, Analog Devices, KEMET, and Texas Instruments.
Is energy harvesting a smart option?
Whether it’s smart or not, of course, depends on the application. For sure, energy harvesting is an attractive and widely used technique for providing power at what at first seems to be little or no cost. Its four key elements are:
- Energy generation (source)
- Conversion (using a transducer of some kind)
- Storage (for use at a later time)
- Distribution (delivering it efficiently to the required circuits)
The first harvesting challenge is to identify the most viable energy source to be tapped. Common sources include solar, wind, hydro, thermal differential, waste heat, or magnetic induction. Another source is some form of minute vibration or motion, which itself can be induced by walking, building geo-vibration, or motor vibration.
It’s important to keep in mind that while the terms energy and power are often used interchangeably, particularly by the general public, they are different, though related. Energy is the ability to do work, while power is the rate at which energy is collected or expended. Therefore, energy is the time integral of power, while power is the time derivative of energy. In a harvesting system, the amount of collected and stored energy must be equal to or greater than its power integral, or the system will not have enough power to function long-term. In other words, energy can’t be expended in the aggregate as power at a higher rate than it is collected over time.
Once the energy source to be harvested is identified, a transducer is needed to capture and transform it into electrical energy. This transducer can take many forms: a microturbine, a solar cell (or panel), or a piezoelectric crystal are just a few possibilities. The next step is to decide how to store this erratic, often unpredictable, and usually small amount of energy so it can be drawn from as needed to power the electronics.
Battery, supercapacitor, or both?
The two viable storage options are the rechargeable battery or the supercapacitor, sometimes called an ultracapacitor, although its formal name is an electric double-layer capacitor (EDLC). Whether to use a rechargeable battery or a supercapacitor is determined largely by the magnitude, duration, and duty cycle of the application.
In general, standard (non-super) capacitors can deliver large amounts of power but store only a relatively small amount of energy per unit volume. In contrast, batteries can store larger amounts of energy, but have lower power ratings. Between the two are supercapacitors, which strike a balance between energy versus power.
Compared to batteries, supercapacitors have a number of advantages:
- Supercapacitors use physical charge storage rather than a chemical reaction, so they can be charged and discharged very quickly (milliseconds to seconds)
- Unlike batteries, the charge-discharge cycle life of a supercapacitor is almost unlimited as it doesn’t rely upon chemical reactions
- Charge management of supercapacitors is simpler than it is for batteries, as they require only charging current and overvoltage protection (OVP) rather than the somewhat complex and chemistry-dependent constant current and constant voltage charging algorithms associated with batteries
For example, the KEMET FC0V474ZFTBR24 is a 47 millifarad (mF) supercapacitor in a small, sealed can measuring 10.5 millimeters (mm) high and 8.5 mm in diameter, with a 3.5 volt nominal rating (Figure 1). In the unlikely event of seal leak, only harmless water vapor (a gas) is emitted as the result of the phase-state transition of the liquid water in the electrolyte (diluted sulfuric acid) to gas.
 Figure 1: The model FC0V474ZFTBR24 supercapacitor from KEMET is a 3.5 volt, 47 mF unit in a sealed can just 10.5 mm high and 8.5 mm in diameter. (Image source: KEMET Corp.)
Figure 1: The model FC0V474ZFTBR24 supercapacitor from KEMET is a 3.5 volt, 47 mF unit in a sealed can just 10.5 mm high and 8.5 mm in diameter. (Image source: KEMET Corp.)
Rechargeable batteries can be used in some longer-term situations and are best for applications where the current drain is low but relatively constant, without high peaks and with low duty cycles. Some designs use a pairing of a rechargeable battery with a supercapacitor, with the battery trickle-charging the capacitor, which in turn provides peak load current.
For most longer term applications, lithium-ion (Li-ion) chemistries are the best choice among the many available standard chemistries with respect to electrical performance attributes, as well as energy density by volume and by weight.
However, there are important differences among the various lithium chemistries related to attributes such as output voltage, charge/discharge characteristics, voltage versus remaining capacity, operating temperature range, and the number of charge/discharge cycles. The latter, which like other battery specifications, is also dependent on the depth of discharge per cycle. A high-level summary of key characteristics of two widely used secondary battery chemistries – nickel-cadmium (NiCad, or NiCd) and Li-ion – as well as two basic bulk capacitor types, is shown in Table 1.
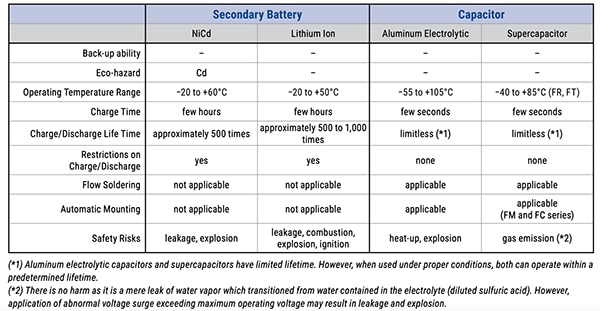 Table 1: Rechargeable batteries and capacitors have different combinations of broad top-tier attributes. (Image source: KEMET Corp.)
Table 1: Rechargeable batteries and capacitors have different combinations of broad top-tier attributes. (Image source: KEMET Corp.)
It’s simple in concept but difficult in practice to determine the needed milliamp-hour (mAh) capacity rating of the energy storage element for applications requiring extremely long duration. While the first-level analysis is based on the integral of the needed power including quiescent, steady state, and pulsed modes, there is more to think about when designing with these devices. Issues such as losses due to internal equivalent series resistance (ESR) and temperature related degradation, are but two of the various factors that must be considered. As a result, careful study of the battery or supercapacitor data sheet and its numerous graphs is required.
System power management: an ongoing challenge
Regardless of whether a battery, supercapacitor, or both in tandem is chosen, management of the energy flow from the harvesting transducer to the storage element(s) and then its delivery to the load is a critical design issue. This function must ensure that the harvested energy, usually a very small value, is transferred to the storage element with maximum efficiency, while at the same time not shortening cell life through overcharging. The management function must also meter current as needed to the load while consuming very little power itself, and it must manage the discharge cycle to avoid deep discharge, which degrades the number of full capacity battery charge/discharge cycles.
On the output side, the manager must also implement DC/DC regulation, so the load rail stays at a constant voltage despite variations in storage element voltage and load demands. Depending upon the battery or capacitor chosen, in the context of the load requirements, this regulation can be a buck or boost mode function. It can also be a combined buck/boost regulator when the storage element output voltage crosses from being above the desired DC rail to below it.
For example, Analog Devices’ LTC3331EUH#PBF is a nanopower buck/boost DC/DC regulator with an energy harvesting battery charger that is optimized for the higher source voltages of solar cells (Figure 2). However, it can also be used with lower voltage sources to form the foundation of a complete energy harvesting solution with battery backup. Housed in a tiny 5 mm × 5mm QFN-32 package, the LTC3331 features dual inputs (for solar and piezo as harvest sources, for example), and a single-output DC/DC regulator which prioritizes between the two inputs.
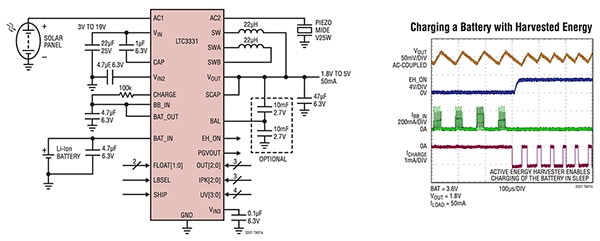 Figure 2: The LTC3331 from Analog Devices can handle and prioritize between two harvesting sources and balance two supercapacitors plus a battery, while delivering between 1.8 volts and 5 volts at up to 50 mA (left). The timing sequence of the LTC3331 when charging a battery from harvested energy is also shown (right). (Image source: Analog Devices)
Figure 2: The LTC3331 from Analog Devices can handle and prioritize between two harvesting sources and balance two supercapacitors plus a battery, while delivering between 1.8 volts and 5 volts at up to 50 mA (left). The timing sequence of the LTC3331 when charging a battery from harvested energy is also shown (right). (Image source: Analog Devices)
The harvested inputs can be between 3.0 volts and 19 volts, while the battery voltage can range up to 4.2 volts; the regulated output rail can be set to between 1.8 volts and 5 volts at 50 mA. It also supports use of two supercapacitors in series, which is done to increase energy storage and efficiency through the use of automatic cell balancing.
The buck and boost converters are controlled by the prioritizer. This selects the source to use based on the availability of a battery and/or harvestable energy. If harvested energy is available, the buck regulator is active and the buck-boost is off. An onboard 10 mA shunt battery charger with low battery disconnect enables charging of the backup battery to greatly extend its life. Quiescent current, which is a critical factor in regulator efficiency, and especially so in harvesting designs, is just 950 nA at no load.
Long-term supply problem solved – or maybe not
If it seems that decisions about available harvest source, use of battery or supercapacitor, and system tradeoffs are complicated, they can be. But there are two bigger issues when using harvesting as the energy source, especially with batteries. First, rechargeable batteries do not have an infinite number of charge/discharge cycles. Their typical 1000 or 2000 cycle rating, even under optimal charge/discharge conditions and temperature, falls short of what’s required over decades.
Second, there’s a larger, hard to quantify question related to the energy source and transducer. Will the solar cell really be fully illuminated for decades in the face of dirt, dust, or someone building something nearby and blocking the light, or trees growing and shadowing the cells? Will the vibration source always be getting stimulation, even as its settings and operating environment changes? These are very difficult issues to address and confirm with a high degree of confidence in many real-world installations.
With regard to storage, it’s also somewhat counterintuitive that a non-rechargeable primary cell battery would even be considered for multidecade endurance, even if it has a microamp or low milliamp load; after all, wouldn’t it dry out, corrode, or suffer some other long-term related malady? However, when using the right type of primary battery under the right conditions, this is actually a very viable alternative to harvesting. In addition, by definition, using primary cells completely avoids all the issues associated with charge/discharge cycles and management.
That said, primary cells do suffer from an unavoidable and deteriorating phenomenon called self-discharge in which there is a small but definite amount of internal leakage current, even if the battery has no load or is physically disconnected. This self-discharge for most types of primary lithium-based cells is on the order of 3 to 4% of initial capacity per year.
Basic math shows that with self-discharge of 4%, the cell capacity will degrade down to about half its original value in about 12 years, not counting any loss of capacity due to supporting the load. As a result of self-discharge, determining lifetime using a simple calculation of load current drain versus initial battery capacity is wildly optimistic, and frankly unrealistic. For that reason, most primary cells are not suitable for long-term, multidecade applications.
However, batteries based on a lithium thionyl chloride (LiSOCl2) chemistry that use a bobbin-type internal arrangement and proprietary manufacturing processes can last for decades, without excessive self-discharge. When used at the very low current levels of many circuits, combined with their self-discharge below 1%, they can power a system for two, three, and even four decades (Figure 3). They are also light in weight but dense in capacity: a high-performance LiSOCl2 cell offers energy density by weight of ~650 watt hours per kilogram (W-hr/kg) and by volume of ~1280 W-hr/cubic decimeter.
Per Figure 3, Tadiran’s LiSOCl2 XOL Series delivers 86% of rated capacity after 20 years (left). Its LiSOCl2 XTRA Series is at 80% after 10 years, while other chemistries are down to 70% (center). The high annual self-discharge rates of lithium manganese dioxide (LMNO2) and alkaline cells make 10+ year battery life unreachable (right).
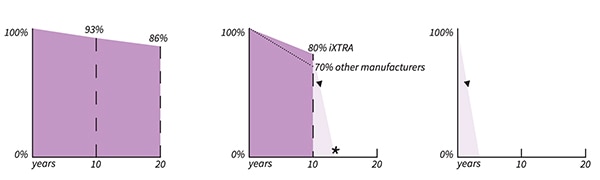 Figure 3: Battery capacity for three different chemistries after 10 and 20 years solely due to self-discharge loss (at no load). Tadiran’s LiSOCl2 XOL Series delivers 86% of rated capacity after 20 years (left); Tadiran’s LiSOCl2 XTRA Series is at 80% after 10 years, while other chemistries are down to 70% (center). The high annual self-discharge rates of LMNO2 and alkaline cells make 10+ year battery life unreachable (right). (Image source: Tadiran Batteries)
Figure 3: Battery capacity for three different chemistries after 10 and 20 years solely due to self-discharge loss (at no load). Tadiran’s LiSOCl2 XOL Series delivers 86% of rated capacity after 20 years (left); Tadiran’s LiSOCl2 XTRA Series is at 80% after 10 years, while other chemistries are down to 70% (center). The high annual self-discharge rates of LMNO2 and alkaline cells make 10+ year battery life unreachable (right). (Image source: Tadiran Batteries)
The key to a LiSOCL2’s low self-discharge is a lithium-chloride (LiCl) passivation layer that inherently forms on the surface of the anode as soon as the lithium comes into contact with the electrolyte, thereby preventing further reaction or loss of capacity. It is an imperfect insulator which greatly limits the flow of self-discharge current, but it is partially “pushed aside” by the low-level current when the load calls for it.
In theory, the passivation layer’s thickness can be increased by design and fabrication to further limit the self-discharge, but then the cell would not work well at higher load currents. So, there’s a trade-off: accept low self-discharge but restrict use to low drain applications, or increase self-discharge further but use the cell at a higher drain for less time (for a given capacity rating).
There are cells available that are optimized for ultra-low drain performance. Consider the TL-4902/S ½ AA size LiSOCl2 primary cell from the Tadiran XLO series, which is just 25 mm long with diameter of 14.5 mm. This cylindrical battery has a terminal voltage of 3.6 volts, with nominal capacity of 1.2 Ah at 0.5 mA down to 2 volts (Figure 4). The output voltage is flat over time, which increases dramatically with lower drain levels, reaching 100,000 hours at 10 microamps (µA).
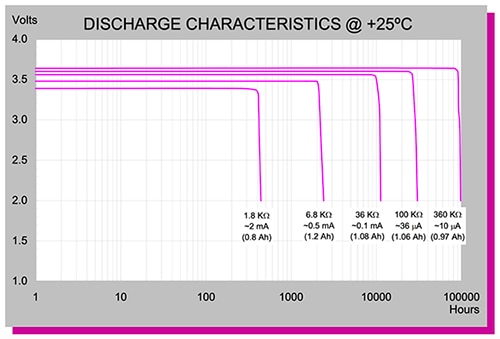 Figure 4: The Tadiran TL-4902/S LiSOCl2 primary battery can supply 10 µA for 100,000 hours with no decrease in output voltage. (Image source: Tadiran Batteries)
Figure 4: The Tadiran TL-4902/S LiSOCl2 primary battery can supply 10 µA for 100,000 hours with no decrease in output voltage. (Image source: Tadiran Batteries)
What about pulsed current applications?
Note that “cutting through” the passivation layer takes on the order of milliseconds, so when the load circuit calls for current there is in reality a transient voltage drop, followed by a slow rise to the nominal output voltage. Therefore, to maximize the long life of which these cells are capable, they should not be used for pulsed loads but instead serve as continuous low current sources.
That said, there is a convenient and practical solution for pulsed loads: combine the long-life, continuous discharge, low current battery with a supercapacitor. Here, the circuit is configured so that the battery continuously charges the supercapacitor at a low rate, while the supercapacitor is used to deliver the higher pulse current (Figure 5). In effect, the arrangement uses the battery as the energy source to be harvested, although not in the standard use of the term.
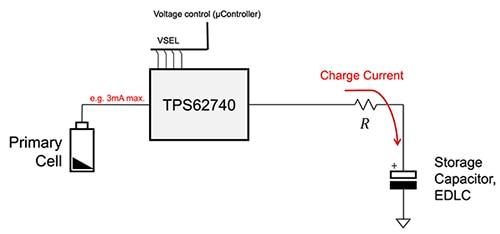 Figure 5: Designers can get ultra-long duration power for pulsed loads in an easily managed arrangement by using a low value, continuous current from a long-life primary cell to trickle-charge a supercapacitor, and then use the supercapacitor to support low duty cycle pulsed loads. (Image source: Texas Instruments)
Figure 5: Designers can get ultra-long duration power for pulsed loads in an easily managed arrangement by using a low value, continuous current from a long-life primary cell to trickle-charge a supercapacitor, and then use the supercapacitor to support low duty cycle pulsed loads. (Image source: Texas Instruments)
The Texas Instruments TPS62740 IC is well-suited to this configuration (Figure 6). This buck converter accepts an input voltage ranging from 2.2 volts to 5.5 volts, has a 360 nA quiescent current specification, and operates with a tiny 2.2 microhenry (µH) inductor and a 10 microfarad (μF) output capacitor while delivering up to 300 mA.
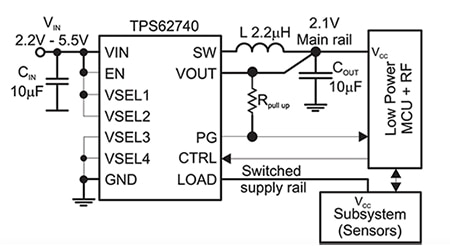 Figure 6: The Texas Instruments TPS62740 step-down converter allows use of a battery to charge the small capacitor and so allows designers to make optimal use of the attributes of each energy storage device. (Image source: Texas Instruments)
Figure 6: The Texas Instruments TPS62740 step-down converter allows use of a battery to charge the small capacitor and so allows designers to make optimal use of the attributes of each energy storage device. (Image source: Texas Instruments)
In a typical application, the LiSOCl2 primary cell is connected directly to the TPS62740 and is controlled by a microcontroller which enables/disables the buck converter, adjusts the output voltage, and enables efficient charging (Figure 7).
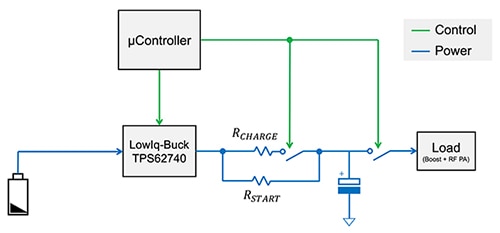 Figure 7: Combining the TPS62740 with a LiSOCl2 primary cell and capacitor with direction by a microcontroller, results in an efficient, long duration power subsystem with low quiescent current operation. (Image source: Texas Instruments)
Figure 7: Combining the TPS62740 with a LiSOCl2 primary cell and capacitor with direction by a microcontroller, results in an efficient, long duration power subsystem with low quiescent current operation. (Image source: Texas Instruments)
The buck converter output is connected to two current limiting resistors to handle the start-up process, which is necessary to pre-charge the supercapacitor to its 1.9 volt minimum. Once the storage capacitor is pre-charged, the switch is turned on, and the current is limited by the combined resistance. With this arrangement, a low duty cycle, large peak current load, such as a wireless IoT node, can be supported with maximum battery life and multidecade operating lifetime.
There are three advantages to using the primary cell approach rather than harvesting with a rechargeable battery, even with an added supercapacitor for pulse current buffering:
- Elimination of the cost of the harvesting transducer itself and the uncertainties of its long-term viability
- Elimination of battery management issues related to the limited number of charge/discharge cycles as well as their dependence on discharge depth and operating temperature
- Simplification of the power management subsystem
Conclusion
Devising a power supply which will provide energy for decades without attention or intervention is a challenging requirement. That’s true even for the relatively modest requirements of the ultra-low current and power designs used for many sensor-based IoT applications.
Use of a low passivation LiSOCl2 primary battery alone for low-current applications, or that battery chemistry in combination with a supercapacitor for pulsed, low duty cycle loads, offers an excellent alternative to the more obvious, perhaps more intuitive choice of energy harvesting with a rechargeable battery.

Disclaimer: The opinions, beliefs, and viewpoints expressed by the various authors and/or forum participants on this website do not necessarily reflect the opinions, beliefs, and viewpoints of DigiKey or official policies of DigiKey.





